Project Charter
The Pistoia Alliance is developing an advanced semantic architecture for a Pharmaceutical CMC Process Ontology. The objective is to create a domain lexicon and taxonomy that extend the ISA‑88 framework, enabling standardized laboratory and plant production process recipes.
This standardization establishes consistent definitions, supports digital technology transfer, and enables integration with execution systems to capture structured process data. As a result, the ontology improves material lot genealogy tracking, streamlines technology transfer, and enables advanced process analytics, enhancing efficiency and transparency across the pharmaceutical production lifecycle.
During the initial project phase (2024), the team delivered significant gains in addressing inefficiencies in technology transfer and data integration across stages and scales, as well as integration across production systems. We showed how an ontology‑driven approach can improve efficiency and transparency throughout the pharmaceutical production lifecycle. In the second phase (2025), we expanded the ontology scope to facilitate optimal process scale-up. The ontology now captures and defines process steps at the development stage of chemical, monoclonal antibody (mAb), and CAR‑T manufacturing, including key process parameters in the P-S-O-A (process, stage, operation, action) model.
Phase 3 (2026) will focus on three complementary areas to ensure both near-term impact and long-term sustainability:
- Cross-domain connectivity and interoperability
- Practical implementation and real-world enablement
- Expansion of the ontology coverage
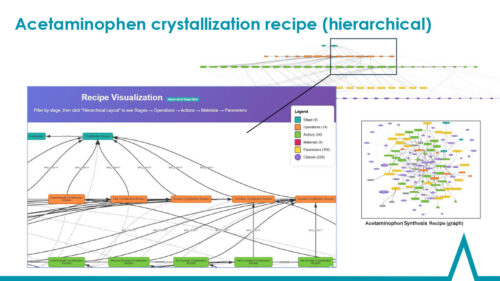
We are building out integration among the CMC Process, Equipment, Material, Analytical (measurement and quality attribute), and IDMP ontologies to ensure interoperability across the value chain. The Pistoia Alliance’s CMC Process Ontology imports the IOF Biopharma Ontologies to support aspects of biopharmaceutical CMC, and is aligned with the Basic Formal Ontology to facilitate broad interoperability.
Get involved in Phase 3
Talk to Birthe Nielsen to learn more and get involved in Phase 2 of this project
Contact Birthe





























